His achievements reshaped modern physics
Hans Dehmelt, the experimental physicist whose meticulous work made it possible to isolate and measure a single electron, has died at the age of 94. Awarded the Nobel Prize in Physics in 1989, Dehmelt helped transform atomic physics by proving that even the smallest charged particles could be trapped, observed, and studied with astonishing precision.
His achievements reshaped modern physics. At a time when atoms and subatomic particles were still largely explored through indirect measurements, Dehmelt demonstrated that individual electrons could be confined long enough to probe their fundamental properties. His work not only deepened scientific understanding but also laid technical foundations for advances in quantum measurement, precision timekeeping, and experimental particle physics.
A Prize for Precision
In 1989, Dehmelt shared the Nobel Prize with Wolfgang Paul and Norman F. Ramsey. The award recognized the development of ion trap techniques and precision measurement methods that allowed scientists to isolate charged particles and examine them in unprecedented detail.
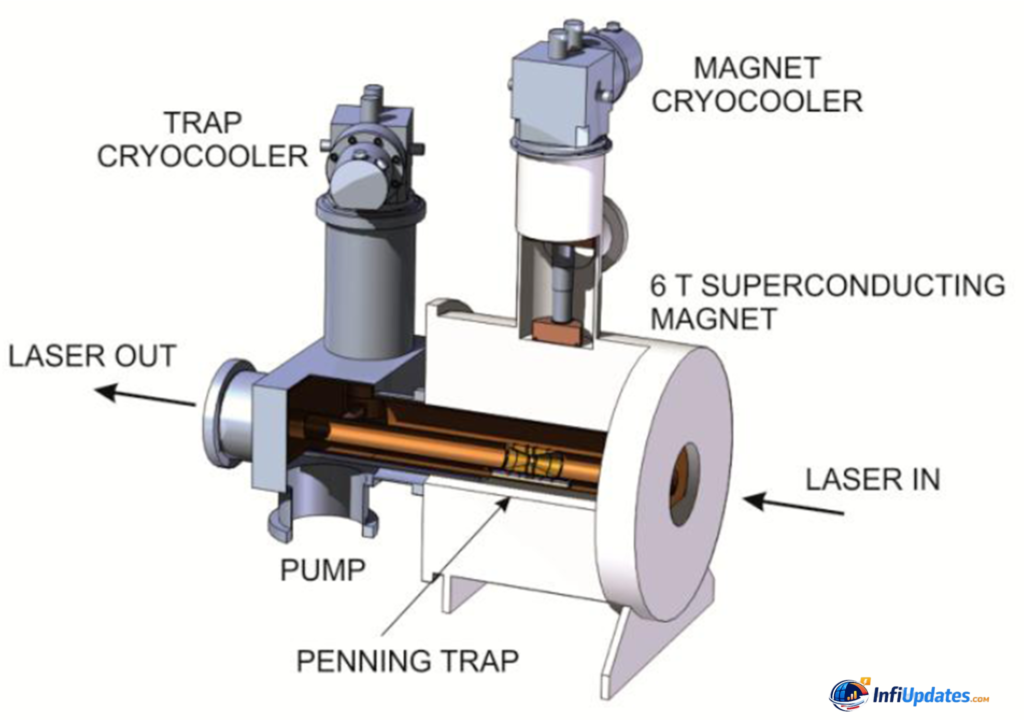
At the center of Dehmelt’s work was the refinement of the Penning trap, a device that uses a combination of magnetic and electric fields to confine charged particles in a small region of space. By carefully controlling these fields, Dehmelt and his team were able to hold a single electron virtually motionless for extended periods. This allowed them to measure the electron’s magnetic moment — a key property that tests the accuracy of quantum electrodynamics (QED), one of the most successful theories in physics.
The results were stunning. The measurements matched theoretical predictions with extraordinary accuracy, confirming QED to a precision rarely achieved in science. In doing so, Dehmelt strengthened confidence in the mathematical framework that describes how light and matter interact.
From Germany to America
Born in 1922 in Görlitz, Germany, Dehmelt came of age during a turbulent era in European history. After serving in the German military during World War II, he pursued his academic ambitions with determination. He later moved to the United States, where he completed his doctorate at Duke University and eventually became a professor at the University of Washington.

At the University of Washington, Dehmelt built a laboratory that became internationally known for precision measurement. Colleagues and students recall his intense focus, dry wit, and relentless pursuit of accuracy. He often described his experimental philosophy as one of patience and persistence — improving apparatus step by step until nature revealed its secrets.
Isolating the Electron
The electron, discovered in the late 19th century, is one of the fundamental building blocks of matter. Yet isolating a single electron for detailed study posed enormous technical challenges. Charged particles are easily disturbed by stray electric fields, vibrations, and even minute temperature fluctuations.
Dehmelt’s genius lay in reducing these disturbances to nearly zero. By cooling particles and fine-tuning electromagnetic traps, he created conditions in which a lone electron could remain confined for months. In some experiments, the particle was observed for nearly a year.
This achievement was more than symbolic. Studying a single particle eliminates the statistical noise that arises when measuring large groups. It allowed for unprecedented experimental precision and set new standards for laboratory physics.
A Legacy Beyond the Laboratory
The technologies refined in Dehmelt’s lab have influenced many fields. Ion trapping techniques now play a role in developing quantum computing systems, atomic clocks, and high-precision spectroscopy. The ability to manipulate single particles has become central to emerging quantum technologies.
Dehmelt also inspired generations of physicists. His laboratory was known for encouraging creative thinking while maintaining uncompromising standards of accuracy. Students who trained under him went on to lead major research programs around the world.
Despite his achievements, Dehmelt remained modest. He often credited collaborators and emphasized that progress in physics comes from collective effort. The Nobel Prize, he once remarked, recognized decades of incremental improvement rather than a single dramatic breakthrough.
A Quiet Revolutionary
Unlike some scientific breakthroughs that capture immediate public imagination, Dehmelt’s work unfolded quietly. There were no dramatic explosions or visible spectacles — only careful measurements, refined instruments, and pages of data. Yet the implications were profound. By isolating and studying the electron, he helped verify the internal consistency of modern physics at a level of detail once thought impossible.
In an era when theoretical predictions push the boundaries of knowledge, precision experiments like Dehmelt’s provide essential checks. Without such work, physics risks drifting away from empirical grounding. His career stands as a reminder that careful measurement is as vital to discovery as bold ideas.
Remembering Hans Dehmelt
Hans Dehmelt’s passing marks the end of a remarkable chapter in experimental physics. He lived long enough to see his techniques become foundational tools for 21st-century science. What began as an effort to trap a single particle evolved into a broader revolution in how physicists observe and measure the microscopic world.
He leaves behind not only a body of pioneering research but also a legacy of intellectual rigor and scientific curiosity. In isolating the electron, Dehmelt did more than confine a particle — he expanded humanity’s ability to understand the universe at its most fundamental level.
As physicists continue to probe deeper into the quantum realm, the methods he perfected remain central. His life’s work stands as proof that even the smallest particles can illuminate the biggest questions in science.